Uvc placement1/31/2024 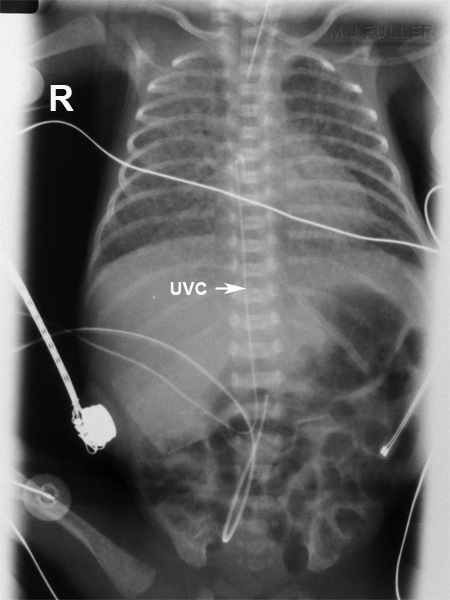 
Notify any customers who may have distributed the affected product and share a copy of the IFU.Share the IFU with all personnel who use the Argyle UVC Insertion Tray containing the Safety Scalpel N11.Review the IFU for the Safety Scalpel N11 that was included with the letter.The letter stated the reason for recall, included the IFU for the Safety Scalpel N11 and provided the following instructions: On June 16, 2021, Cardinal Health sent all affected customers an Urgent Medical Device Correction letter. Patients who have procedures with the Safety Scalpel N11.Health care providers who use the Argyle UVC Insertion Tray containing the Safety Scalpel N11.There were two reports of death in which the Safety Scalpel within the UVC kit locked permanently, and the clinician was unable to unlock for use during emergency UVC placement. There have been 10 complaints reported about this device issue. A clinician’s inability to use the scalpel (when in the permanent locked position) poses a safety risk to the patient that could result in delayed treatment which could lead to serious injury or death. If the clinician is not aware of this permanent locking feature, there could be a delay in the procedure due to obtaining a replacement scalpel.Ĭritically ill newborn patients may require emergency umbilical vein catheter placement. The Safety Scalpel N11 has a permanent locking feature that will not allow a clinician to unlock the scalpel once they have closed the safety shield and pushed it forward into a locked position (Figure 4). 
Figure 4: Safety Scalpel N11 – Closed & Locked Position Reason for RecallĬardinal Health is recalling the Argyle UVC Insertion Tray because it does not include specific Instructions for Use (IFU) for the Safety Scalpel N11 contained in the tray.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
